Kendall Lab
The Kendall Lab is focused on developing basic and translational models of pediatric sarcomas—devastating and aggressive solid tumors with limited therapeutic options. Specifically, we study fusion-driven rhabdomyosarcoma, which is thought to arise from a misregulation of skeletal muscle developmental pathways. We use cell culture and transgenic zebrafish systems to delineate the underlying biology of the disease and then apply that knowledge to identify molecular targets. Our ultimate goal is to apply our models as discovery- based tools to derive mechanistic insights for improving outcomes for children with cancer.
Inside the Kendall Lab
Our Lab Funding
- NIH/NCI R01 CA272872
- Alex’s Lemonade Stand Foundation “A” Award co-funded by Flashes of Hope
- The V Foundation for Cancer Research V Scholar Award
- CancerFree Kids New Idea Award
Kendall Lab in the Media
VIDEO: Inside our Nationwide Children’s Hospital Zebrafish Facility: Genetic, Life-Saving Research
PODCAST: In Episode 75, Quinn & Brian discuss: Beating the crap out of childhood cancer with science and... zebrafish? We’re getting really nerdy about beating childhood cancer with Dr. Jaclyn Taroni and Dr. Genevieve Kendall.
Zebrafish expose pathway in childhood muscle cancer.
- DALLAS – Jun. 5, 2018 – A popular aquarium fish may hold answers to how tumors form in a childhood cancer.
BLOG: A passion for pediatric sarcoma research – AACR Cancer Research Catalyst
- Genevieve Kendall, PhD, has focused her research on this specific form of sarcoma, with the ultimate goal of developing new animal models and applying them to identify active small molecules or treatment strategies that could be implemented to improve patient outcomes.
Join Our Team!
We are very committed to mentoring the next generation of scientists. We are looking for enthusiastic, self-motivated individuals who thrive in a fast-paced challenging start-up research environment. Individuals must be able to work well with others as part of a cohesive team, while also being comfortable producing and pursuing independent ideas. If you have a desire to make a significant impact in pediatric cancers, we invite you to contact us concerning a position.
Featured Research Projects
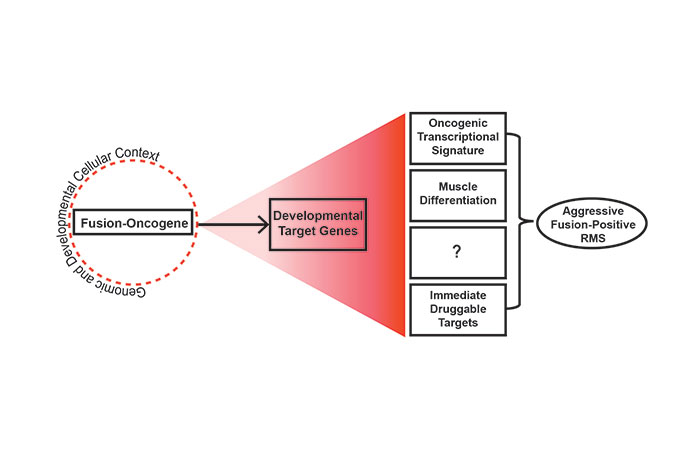
We found that HES3 is a novel oncogenic target of PAX3-FOXO1. My group will now focus on how HES3 and PAX3-FOXO1 cooperate to inhibit developmental programs, such as myogenesis, and the cellular transcriptional response required for transformation. In parallel, to advance timely therapeutics in RMS, we will use chemical and genetic strategies to test a subset of clinically tractable targets identified in the more aggressive HES3-high RMS patient signature.
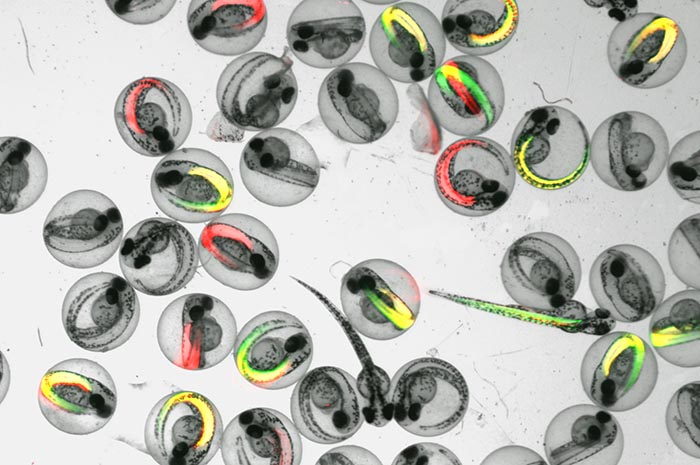
We have implemented genetic strategies to restrict cell lineage specific expression of human fusion-oncogenes in zebrafish systems. This is an ongoing collaboration with Koichi Kawakami at the National Institute of Genetics in Mishima, Japan. Over five years I have visited his lab and collected a unique sarcoma shelf screening resource consisting of over 50 transgenic zebrafish lines. These transgenic lines are being implemented to restrict the expression of human fusion-oncogenes to specific cellular compartments across a spectrum of developmental timepoints. Our efforts now include performing sarcoma shelf-screens to identify cell lineages that are resistant or susceptible to sarcoma fusion-oncogene expression. Further, we will understand the biology of these fusion-oncogenes by defining the genomic and transcriptional features of their in vivo activity. Comparing shared and unique vulnerabilities across multiple fusion-oncogenes will be a key approach for the identification of potential targets.
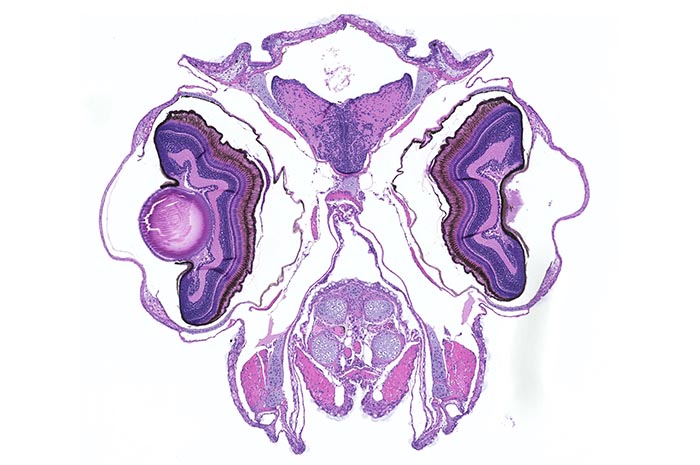
We are well positioned at Nationwide Children’s Hospital, and through multiple collaborations, to intersect with clinical sequencing efforts and validate novel oncogenic drivers. Our group generates new models of pediatric sarcoma by integrating newly identified fusion-oncogenes into the zebrafish genome to functionally validate their transformative capacity. These animal models allow us to understand the biology of these rare diseases in an appropriate developmental context and are a unique platform to identify potential targeted therapies.



