Heart Development and Regeneration Lab
The Heart Development and Regeneration Lab aims to understand the fundamental mechanisms of cardiac cell proliferation and differentiation and how signaling communications instruct these processes:
- How do the cells in the developing heart communicate with each other to build a functional heart?
- How are cardiac cell lineages determined during early heart development?
- How can we promote adult regeneration by remodeling chromatin accessibility?
The lab takes advantage of multiple cutting-edge technologies, including single cell RNA sequencing, ribosome profiling, ChIP-sequencing, CROP-Seq, ATAC-Seq, special transcriptomics, proteomics, CRISPR-Cas9 and confocal imaging, and generates and uses genetic mouse models and human induced pluripotent cells to tackle these and other important questions in the field.
Inside Li Lab
Our Research
Research in my laboratory is focused on understanding the mechanisms of cardiovascular development, disease and regeneration. Heart is composed of at least 10 different types of cells, which are functioned both autonomously and dependently. Any disruptions of these functions can lead to heart malformation or disease. However, their functions are still poorly understood. Filling this knowledge gap will improve our understanding of disease pathogenesis and thus shed lights on novel therapeutics. The lab is using multiple lines of cutting-edge technologies such as genetic mouse models, iPSCs, CRISPR/CAS9, scRNA-seq, to address these critical scientific questions. There are several ongoing exciting research projects in the lab.
Featured Publications
- HDAC7 Promotes Cardiomyocyte Proliferation by Suppressing Myocyte Enhancer Factor 2
- Endocardial HDAC3 is Required for Myocardial Trabeculation
- Epicardial HDAC3 Promotes Myocardial Growth Through a Novel MicroRNA Pathway
Featured Research Projects
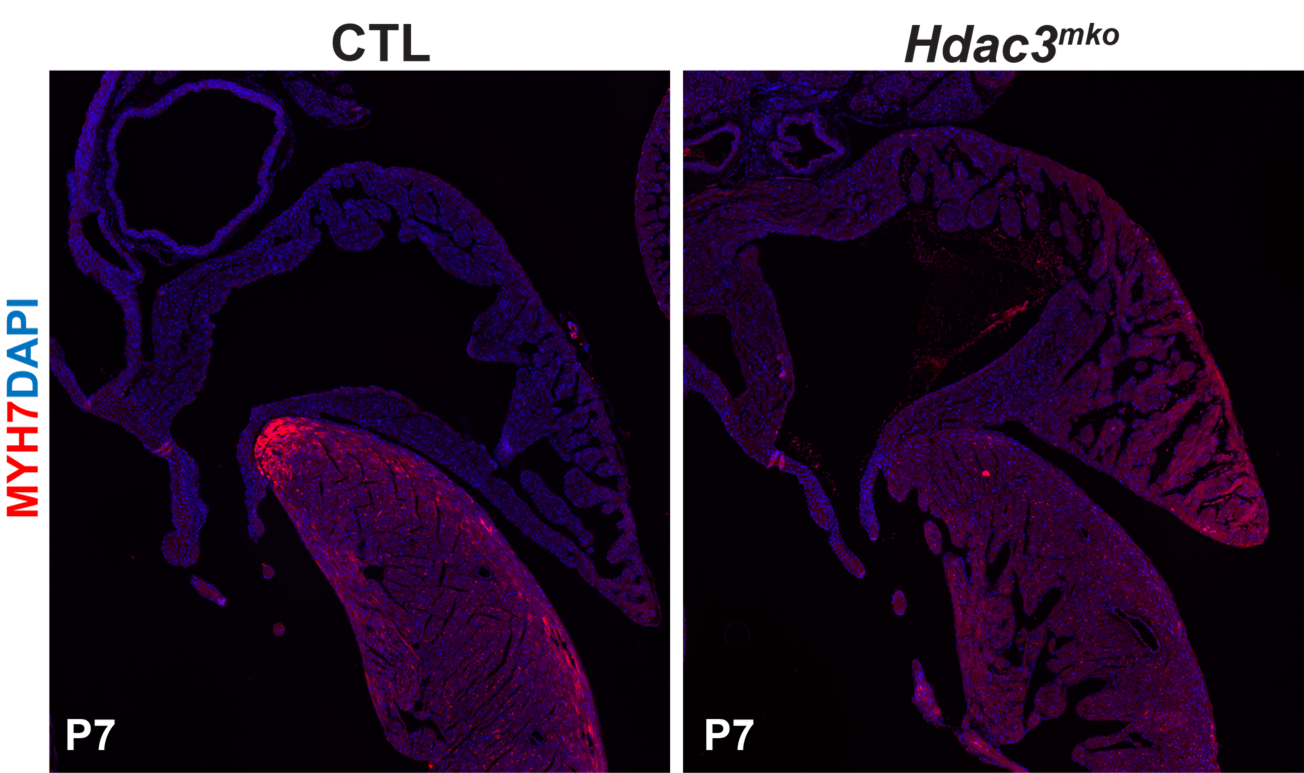
Role of Hdac3 in atrial identity and function. Our preliminary data showed that deletion of Hdac3 in the developing atria in mice results in gaining of ventricular identity. Currently, we are investigating the mechanisms by which Hdac3 establishes/maintains atrial identity.
Role of endocardial Zip8 driving myocardial trabecular morphogenesis. Our preliminary data showed that deletion of Zip8 in the developing endocardium in mice results in a phenotype similar to a clinical condition called “left ventricular noncompaction cardiomyopathy”. Our current findings suggest that endocardial Zip8 accomplishes this by regulating MTF1 transcriptional activities through controlling intracellular zinc availability.
Understanding how hypoxia can impact on cardiac morphogenesis and regeneration. There is increasing body of work suggesting that hypoxia stimulates cardiac regeneration. Our lab is currently investigating whether genetic hypoxia in the endocardium will promote myocardial regeneration.
Join Our Lab
We are committed to training the next generation of scientists as the true future of science and medicine. We are looking for highly motivated postdoctoral researchers and students to join our team. View open positions or contact our team using the form below.



