Tetralogy of Fallot (TOF)
Tetralogy of Fallot is a congenital heart defect that is made up of 4 problems and results in not enough blood flow to the lungs.
What Is Tetralogy of Fallot?
Tetralogy of Fallot (TOF) includes 4 congenital heart problems. With TOF, the lungs do not get enough blood. This condition can be mild to severe.
These 4 heart problems in TOF are:
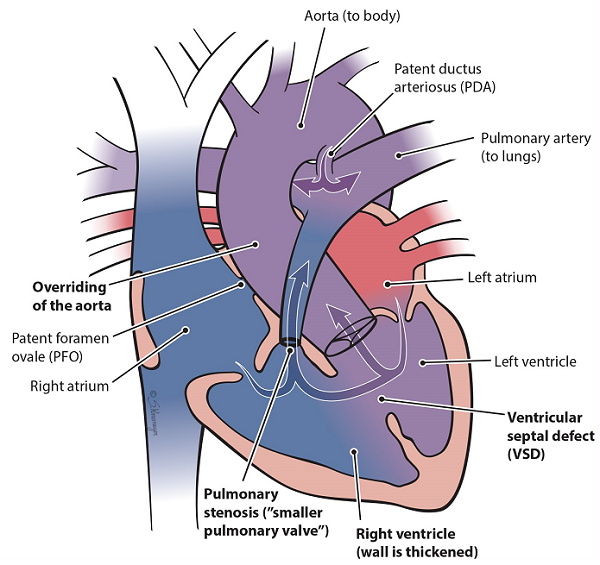
- Ventricular septal defect (VSD): A hole between the 2 bottom pumping chambers of the heart (ventricles).
- Overriding of the aorta: In the normal heart, the aorta comes off the left ventricle. In TOF, the aorta sits between the right and left ventricles, over the VSD.
- Pulmonary stenosis: The pulmonary valve is more narrow than normal and does not open properly. This makes it harder for the right ventricle to pump blood to the lungs. This causes the baby to have low oxygen levels.
- Right ventricular hypertrophy: The right part of the heart has to work harder. This causes the walls of the right pumping chamber (ventricle) to get thicker. This is called hypertrophy.
What Causes Tetralogy of Fallot?
In most cases, parents have no control over their baby’s condition. They have not done anything to cause the problem. In many cases, the cause of congenital heart defects is not known. If you have questions or concerns about you or your baby, talk with your doctor, health care provider, or nurse practitioner.
Diagnosis of Tetralogy of Fallot
When you are pregnant, there are many tests that can tell the doctor if your baby has a heart (cardiac) problem. However, some problems can't be seen until after your baby is born. The following are some prenatal tests that may be done:
- Prenatal ultrasound: Complex heart defects are usually found during a pregnancy (prenatal) ultrasound. This test tells us if your baby's heart is not shaped the right way. If this is found, you will be scheduled for a fetal echocardiogram to get more information.
- Fetal echocardiogram: A fetal echocardiogram is a special ultrasound used by a pediatric heart doctor (cardiologist). This test looks closely at your baby’s heart and surrounding blood vessels. The cardiologist will assess your baby after birth with an echocardiogram to confirm the diagnosis.
- Fetal MRI (Magnetic Resonance Imaging): This type of imaging gives more detailed pictures of your baby’s organs. It may help find problems with the heart or lungs.
After Your Baby Is Born (Postnatal Diagnosis)
- Echocardiogram: An echocardiogram is a special ultrasound used by a pediatric heart doctor (cardiologist) to look closely at your baby’s heart and surrounding blood vessels.
- Heart Catherization: This is a test to measure pressures in the heart. It can also take pictures of the heart and open a narrowing with a balloon or small metal coil, called a stent. Heart catheterizations are done while your child is asleep with medicines called anesthesia.
- Cardiac MRI: A test that uses radio waves, magnets, and a computer to make detailed pictures of the heart and blood vessels. Cardiac MRI can provide detailed information on the type and severity of heart disease.
Other Tests:
Some congenital conditions are caused by a baby’s DNA. The doctors may suggest genetic testing to find out more about this. DNA is made up of information inside your baby’s cells that make them who they are, like blueprints for a house.
- Cell-Free Fetal DNA Testing: A sample of blood is taken to look for copies of your baby's (fetal) DNA. A positive result means that there could be problems with the DNA. This must be confirmed with another test to be sure of the result. Other testing may be done before or after birth, depending on your baby’s needs.
- Amniocentesis: A small sample of amniotic fluid that surrounds the baby in the womb is taken and tested for DNA problems. This test is usually done in the middle of the second trimester of pregnancy, usually between 14 and 20 weeks. It can be used to confirm findings from the cell-free fetal DNA test.
- Karyotype or Microarray: A sample of your baby’s blood is taken to test for DNA problems. This is called either a karyotype (basic chromosome study) or a microarray (more detailed look at all genes).
How Is Tetralogy of Fallot Treated?
At Nationwide Children’s, we provide care for tetralogy of Fallot across all stages of life, from prenatal to adulthood.
All children born with tetralogy of Fallot need to have surgery at some point. Most children will need surgery before they turn 1 year old. This is usually around 6 months of age. The goal is to improve blood flow to the lungs and the rest of the body. Some children will need a temporary shunt placed to provide stable pulmonary blood flow until a more permanent repair can be made at a later age. Surgery will fix the pulmonary stenosis by opening the pulmonary valve and closing the VSD using a patch. Another surgery later in life is sometimes need.
Care During Your Pregnancy
If you are carrying a baby with a heart defect, you will still get care from your pregnancy doctor (obstetrician). They may refer you to a maternal-fetal medicine (MFM) specialist or the Fetal Center at Nationwide Children’s. They will help you connect with a team of fetal experts. At the Fetal Center, you will meet with a multidisciplinary team including cardiologists (doctors who specialize in babies' hearts), neonatologists (doctors who specialize in treating newborns), cardiothoracic surgeons (doctors who perform surgery), and nurses. They will help you learn more about your baby’s care after they are born. Nurse coordinators can answer your questions, guide you through your pregnancy, and prepare you for what to expect.
During clinic visits, the medical team will discuss the diagnosis, treatment, recovery, and long-term outcome for your baby. You will get a tour of the Cardiac Intensive Care Unit so you know what to expect during the hospital stay.
Your doctor will watch your pregnancy closely. You will deliver your baby at a hospital that is ready to care for babies like yours. Talk to your obstetrician about your plans for labor and delivery. Contact your doctor right away if you have any concerns.
Care After Your Baby Is Born
Some heart conditions won’t need to be fixed right away. It can be better to give your baby the chance to grow, making recovery much easier. For these cases, your baby can safely stay close to you at the delivery hospital before they see the heart doctor (cardiologist). The medical team may order tests like an echocardiogram or genetic studies based on your baby’s condition.
Some heart conditions are more severe and can cause problems right away. At the delivery hospital, a doctor who specializes in working with sick infants (neonatologist) will manage the care of your baby. They will watch your baby’s breathing and heart rate and look for other problems. Babies born with heart defects may need care in the Neonatal Intensive Care Unit (NICU) at the delivery hospital. An intravenous line (IV) may be started in your baby’s belly button (umbilicus), arm, leg, or scalp. It will give fluids, medicines, and antibiotics to prevent dehydration and infection.
Your baby may need help with breathing, extra oxygen, or may need to be started on a medicine called prostaglandin E (PGE, or "prostins"). This medicine keeps the blood flowing through the patent ductus arteriosus (PDA). Keeping the PDA open lets blood flow to your child’s lungs or body, helping to keep them stable until surgery.
If your baby’s condition is severe, they will need to be transferred to Nationwide Children’s Hospital Cardiothoracic Intensive Care Unit (CTICU) shortly after birth by the Nationwide Children’s Mobile Intensive Care team. The team is experienced at moving newborns with heart defects. Every effort is made to let you see your baby before they leave the birth hospital.
At Nationwide Children’s Hospital, you will meet with a team of many different specialists. This team includes:
- Cardiologists: Doctors who specialize in working with the heart
- Cardiothoracic surgeons: Doctors who specialize in surgery on the heart and lungs
- Neonatologists: Doctors who specialize in treating newborns
- Interventional cardiologist and catheterization team: A team of health care professionals that specialize in heart procedures
- Other specialized support, like social work, nutrition, therapy, psychology
- A specialized team of nurses and nurse practitioners
Pre-operative Care and Testing (For Surgical Treatment)
When your baby gets to the CTICU at Nationwide Children’s Hospital, they will have many tests. These tests include an echocardiogram and an EKG. These tests will help the team make a plan for your baby at the Heart Center Case Management Conference. This plan includes choices about surgeries, procedures, and timing.
Depending on your baby's heart problem and the timing of surgery, they may need a long-term IV called a peripherally inserted central catheter (PICC). It can give medicines and a form of nutrition called total parenteral nutrition (TPN). Blood can also be drawn from the PICC so your baby doesn’t need to be stuck as often.
Because all of the organs are developing at the same time, it is important to check your baby for other problems. A head ultrasound (HUS) will be done in the first few days after birth to look for any brain problems, such as bleeding. An ultrasound of the kidneys (renal ultrasound) will also be done to make sure they are healthy.
Conserving Blood
The Heart Center at Nationwide Children’s Hospital has special methods to save (conserve) as much blood as possible during surgery. This can decrease the need for a blood transfusion later. These methods include:
- Saving your child’s blood lost during surgery to use after surgery
- Using a shorter cardiopulmonary bypass circuit
- Using the smallest amount of blood needed for lab tests
- Increasing time between lab tests
- Stopping lab tests when possible
Care After Surgery
After surgery, your baby will go to the Cardiothoracic Intensive Care Unit (CTICU). They may be on a breathing machine and have many tubes, wires, and equipment to keep them stable and make sure they are okay. Members of their health care team may make changes to your baby’s medicines and breathing machine often. This will happen the most in the first 24 to 48 hours after surgery.
Other treatments your baby will probably get after surgery are:
- Antibiotic medicine to prevent infection
- Fluids and nutrition through an IV or PICC line
- Heart medicines by IV or by mouth
- Chest drainage tube
- Oxygen
- Pain medicines
- A tube in the nose to the stomach (nasogastric [NG] tube) to keep the stomach empty
- Blood transfusions, when needed
Feeding Your Baby
For all babies, growth and nutrition are an important part of their care. Your baby will be able to eat as soon as they are stable and safe. This may be the first chest/breastfeeding in the delivery room, or it may be after surgery. For all parents interested in giving their own milk, we encourage and support this. Some babies may need to start with small amounts of milk at first and slowly increase. Babies with heart defects may tire easily while feeding or have trouble sucking and swallowing. A temporary feeding tube may be placed into your baby’s nose or mouth, down into the stomach. Some babies will need early nutrition through an IV, called total parenteral nutrition (TPN).
Your medical team will help you decide on the safest feeding approach. Lactation nurses and occupational and speech therapists can work with your baby on oral feeding skills. They can also help you with pumping or direct chest/breastfeeding, when needed. Your baby may go home eating by mouth, but some babies may need help getting the calories needed to grow.
Discharge Planning
Your hospital stay after birth may be a couple of days or as long as weeks to months. This depends on the type of heart disease, surgery, prematurity, genetic syndrome, or other problems.
You and your baby will go home when they can:
- Breathe comfortably and with safe oxygen levels.
- Gain weight on a home-going feeding plan.
- Control their own temperature in a regular baby crib.
The hospital staff will talk with you about your baby’s goals and make sure you and your family are comfortable with their care. As needed, therapists, lactation consultants, and social workers will help with resources and give ideas for continued care. You will need to choose a pediatrician for your baby before going home. The medical team will help you make your first pediatrician appointment. They will also set up appointments for cardiology clinics and any other clinics. Your baby may need heart tests before going home. However, these may be scheduled for a later time after discharge.
Discharge Education
You will need to:
- Do CPR training in the Columbus Blue Jackets Family Resource Center before you leave.
- Be able to give well-baby care, feed your child, and understand the medicines that your baby is taking.
- Know the signs and symptoms to watch for at home and when to call and when to bring your child directly to the hospital.
Tests Before Going Home
- Hearing screen: The state requires that all babies must have a hearing screen before going home for the first time.
- Newborn screening: This test is sometimes called a PKU test. It is a lab test done during the first 24 hours of life. It looks at your baby’s blood for many serious but rare medical conditions. If it is not treated, these conditions can lead to slow growth, developmental disabilities, and possible death. Finding and treating these conditions early may prevent serious problems in the future.
- Hepatitis B vaccine: We encourage all parents to have their baby get the hepatitis B shot (vaccine) before going home for the first time. Your child’s pediatrician will give you the schedule for other vaccines.
- Car seat test: This test makes sure your baby’s heart rate, breathing, and oxygen levels are stable while in the car seat. Your baby will need to sit in the car seat with a monitor on for a set amount of time before going home.
- Cardiac tests: Your baby may have a discharge chest X-ray, echocardiogram, and EKG before being sent home.
Looking Ahead
Your baby will, likely, have pediatrician appointments every 2 months for well-baby checks and immunizations. They will work with the heart team to care for your baby.
Your cardiologist will follow up with your baby’s heart needs even after they are discharged from the hospital.
Future Pregnancies
Most of the time, congenital heart defects don’t happen again with future pregnancies. However, it is recommended that you meet with a geneticist, maternal-fetal medicine doctor (high-risk), or obstetrician before getting pregnant again. Your child’s cardiologist can also help answer questions. They can help you find out if you are at high risk of having another child with a heart defect. They can also talk with you about options and tests.
Next Steps
Care needs change as your child grows. At Nationwide Children’s Hospital, we are committed to providing the best care and quality of life for children of all ages, from in utero to adulthood and beyond.
For Expectant Parents With a Prenatal Diagnosis
Our expert team of fetal medicine specialists is here to guide and support you each step of the way.
For Parents of Children and Newborns
As the leader in pediatric care, The Heart Center offers cardiology services, cardiothoracic surgery, and everything else you would expect to find at a nationally-renowned heart center.
Learn more about The Heart CenterFor Adolescents and Adults
Adolescents and adults with congenital heart disease have different considerations than children. Our program was created to meet the medical and surgical needs of this unique and complex population.
Learn more about the Adolescent & Adult Congenital Heart Disease (ACHD) Program



